Flash Distillation
Introduction
Distillation is a unit operation or a physical separation process of separate more volatile components from a mixture by partial vaporisation of the liquid mixture followed by condensation. The distribution of the components in the two phases is governed by the vapour liquid equilibrium relationship.
The most elementary form of simple distillation is Flash Distillation, in which a feed mixture is partially vaporized to produce vapour which is enriched with more volatile components and a liquid that is devoid of volatile components. The vapour and liquid streams leaving the drum are in equilibrium. This operation is called equilibrium flash vaporisation or flash distillation.
VAPOUR-LIQUID EQUILIBRIUM (VLE)
Separation of a mixture by distillation is based on the equilibrium distribution of the components between the liquid phase and the vapour phase. Knowledge of vapour-liquid equilibrium is therefore essential for understanding the principles of distillation. A phase equilibrium diagram is essential for the relationship between the mole fraction of a component in the vapour and liquid phases. The equilibrium is defined for varying temperatures at constant pressure or varying pressure at constant temperature.
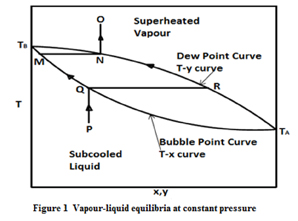
Figure.1 is the phase-equilibrium diagram for a binary mixture system where x, y refers to the mole fraction of the more volatile component in the liquid phase and vapour phase respectively. The temperature raised to its boiling point and is plotted with respect to the liquid composition x, then we get the "bubble point curve" and if we plot it again with respect to the vapour composition y then we get the "dew point curve". In the diagram P represents the sub-cooled liquid which is heated at constant pressure to point Q, where the first bubble of vapour appears. The vapour appeared has the composition, represented by point R which is significantly rich with the less volatile component than in the initial state. The heating process is continued beyond point Q where the temperature rises causing the amount of vapour to increase and the amount of liquid to decrease. During this process, compositions of vapour and liquid phases change as indicated by paths RN and QM until the dew point is reached at point N,where the last droplet of liquid disappear. The line TB-M-Q-TA in the figure.1 is the bubble point curve and TB-N-R-TA is the dew point curve.
RAOULT'S LAW-Raoult's law describes the vapour liquid equilibrium of ideal solution. For an ideal solution the partial pressure of a component is equal to the product of itsmole fraction in the liquid phase and its vapour pressure at the prevailing conditions. It can be written as
- PA* = xAPA
- PB* = xBPB
- where,
- PA*, PB*= Partial pressure of component A and B
- xA, xB = Mole fraction of component A and B in the liquid phase
- PA, PB = Vapour pressures of component A and B at system temperature
- Also,
- PA*= yAP
- PB*= yBP
- Where yA, yB = Mole fraction of component A and B in the vapour phase
- P = Total pressure
According to Dalton's law the total pressure is equal to the sum of the partial pressures. Therefore,
- P =PA*+PB*
- = xAPA+xBPB
The vapour pressure of pure components can be obtained either from charts or the use of equation. Antoine equation given below is widely used for this. This is given below
- In Pi= Ai - Bi/ Ci + T
- Where Ai, Bi and Ci are constant for component i.
RELATIVE VOLATILITY :Relative volatility is the concentration ratio of A to B in the vapour phase divided by the same ratio in the liquid phase. Relative volatility is usually denoted as α. For binary mixture
- α = y/(1-y) / x(1-x)
- PA/ PB (Ratio of vapour pressure of A and B)
α is a dimensionless quantity. When α = 1, the separation would be impossible under the given condition because the composition of the liquid and the vapour phase are the same (azeotrope).When α > 1, separation by distillation becomes progressively easier.
DIFFERENT WAYS OF FLASH DISTILLATION:
The separation of a feed mixture in a flash drum can be carried out by two different ways (Luyben, 1990), and they are described as follows
- The temperature of the liquid feed is increased to a certain temperature which lies between the bubble point temperature and the dew point temperature of that feed at a specified constant pressure,
-
By lowering the pressure on the feed stream.
The partial vaporisation of a multicomponent feed mixture is commonly conducted by reducing the pressure on the feed stream rather than by heating the feed at constant pressure. However, the two general methods referred for flash calculation are as follows:- Isothermal flash
- Adiabatic flash.
ISOTHERMAL FLASH
The isothermal flash is commonly used in the single stage separation process.The flash temperature TF and pressure P. The total flow rate F and composition(xi) of the feed. The name given as isothermal flash because the temperature of the contents in the flash drum as well as the vapour and liquid streams formed by the flash are the same temperature. The flash temperature may not be equal to the feed temperature prior to its flashing. An isothermal flash process can be mathematically described by the independent equation required to describe by the following equation.

Equilibrium relationship
- yFi=KFixFi ( i= 1,2,.....C)
-
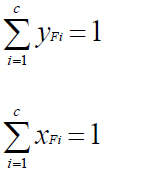
-
Material balances:
FZFi = VFyFi + LFxFi - Where,KFi represents the equilibrium coefficient of component i, yFi and xFi are the composition of component i in the produced vapour and liquid streams.
- ZFi the feed composition of component i, LF and VF the flow rates of liquid and vapour streams. Equation 13 can be extended to:
-
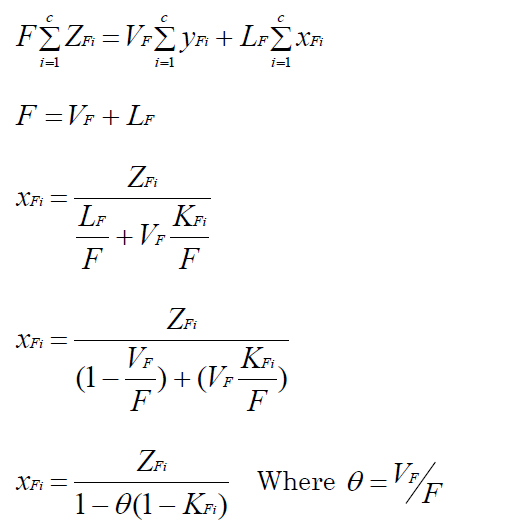
- Now, we consider,
-
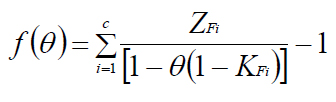
- Positive value of θ means the vaporisation occurs, whereas zero value indicatesno vaporisation.
MULTIPLE CHOICE QUESTIONS
1. Distillation is particularly important in
- Manufacture of sulphuric acid
- Petroleum refinery
- Both (a) and (b)
- None of these
2. Relative volatility, α is given by
- α=y(1-x)/(1-y)x
- α=yx/(1-y)(1-x)
- α=y(1-y)/x(1-x)
- α=x(1-x)/y(1-y)
3. Flash distillation is
- Same as differential distillation
- Used for multicomponent systems like crude refinery
- Same as simple distillation
- Most useful for handling binary system
4. Relative volatility does not change appreciably with the change in
- Temperature
- Total pressure
- Vapour pressure
- None of these
5. The relative volatility
- is independent of pressure
- Increases with increasing pressure
- Decreases with increasing pressure
- Increases with increasing pressure and temperature.
6. Separation of components by distillation is not possible for
- α=1
- α>1
- α=0
- α<1
7. In distillation separation is accomplished by application of
- Solvents
- Heat
- Absorbent
- None of these
8. Roult's law is applicable to
- Ideal solution
- Nonideal solution
- Real solutions
- Both a and b
9. A system comprising of two insoluble liquids, A and B will boil when the total pressure Pt is equal to
- xAsatpA+xBsatPb
- PsatA + satPB
- yAsatPA + yBsatPB
- none of these
10.For a multicomponent system the bubble point is that temperature when
- c∑i=1yi=1
- c∑i=1xi=1
- c∑i=1yi=0
- c∑i=1xi=0
11.For a multicomponent system the dew point is that temperature when
- c∑i=1yi=1
- c∑i=1xi=1
- c∑i=1yi=0
- c∑i=1xi=0
12.For ideal solutions relative volatility, α is related to the vapour pressure of components in a binary mixture according to the relation
- α= PA + PB
- α= PA / PB
- α= PB / PA
- α= PAPB
13.In flash vaporisation,the product vapours
- is always in equilibrium with the liquid leaving the flash chamber
- is never in equilibrium with the liquid leaving the flash chamber
- May be in equilibrium with the liquid leaving the flash chamber and the extent of equilibrium depends on the vapour- liquid contact time in the chamber
- None of these.
Numerical Problems
1. A mixture of 45mol% benzene and 55mol% toluene is being flash distilled at a rate of 15 kmol/hr at 1 atm total pressure. Calculate the amounts and composition of the top and bottom products if the liquid product should not contain more than 35 mol% benzene. The relative volatility of benzene in this mixture is 2.6.
2.Using Antoine equation calculate the bubble point of an ideal solution containing 40 mol% benzene and 60 mol% toluene at 1 atm total pressure. A, B and C for Benzene are respectively 15.9037, 2789.01 and 220.79 and A, B, C for Toluene are respectively 16.00531, 3090.78 and C=219.4.
3.Using Antoine equation calculate the dew point of a vapour containing 65 mol% benzene and 35 mol% toluene at 1.5 bar total pressure .Where A=15.9037,B=2789.01 and C=220.79 for benzene and A=16.00531,B=3090.78 and C=219.4 for toluene.
4.A mixture of 35 mol% n-hexane, 30 mol% n-heptanes and 35 mol% n-octane at 350K and 1.5 atm. total pressure. Will the mixture separate into two phases? If so,calculate the amounts and the vapour products .Assuming the solution to be ideal.
5.An equimolar mixture of benzene and toluene is subjected to flash distillation at a pressure of 1 bar in the separator. Determine the compositions (in mole fraction benzene) of the liquid and vapour leaving the separator when the feed is 30% vaporised. Estimate the temperature in the separator. Equilibrium data for benzene-toluene system at 1 bar is given in below the table
What are the concentrations in the vapour and liquid, and the separator temperature the feed is:
- 0% vaporised
- 50% vaporised
- 75%vaporised
- 100%vaporised
Plot the operating lines for each of these cases and discuss how the operating lines change as f changes.
Virtual Lab Feedback :
1. Amiya K. Jana, "Chemical Process Modelling and Computer Simulation", Prentice-Hall of India Private Limited, New Delhi (2008).
2. Binay K.Dutta, "Principles of Mass Transfer and Separation Processes", Prentice-Hall of India Private Limited, New Delhi (2007).
3. Robert E. Treybal," Mass Transfer Operation", McGraw-Hill Book Company (1981).
4. W.L.Luyben," Process Modelling, Simulation, and Control for Chemical engineers", McGraw-Hill Publishing Company, Singapore (1981).
5. Warren L.McCabe,Julian C. Smith,Peter Harriot, "Unit Operation of Chemical Engineering", McGraw-Hill Companies, New York, International Edition(2005).



